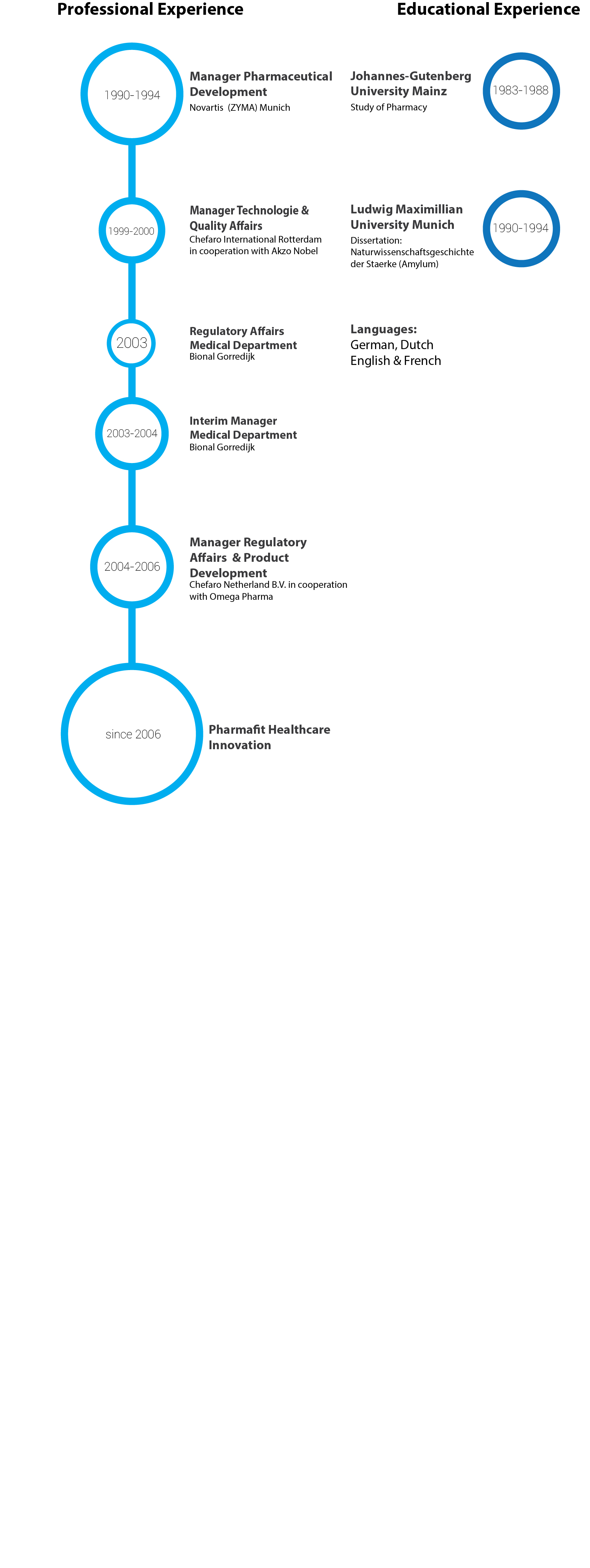
Dr. Mueller-Sarwin started her career in both public and hospital pharmacies in Germany and then she moved on to Pharmaceutical Industry in 1990 in Germany and the Netherlands. Her key expertise area includes Regulatory Affairs CMC (Chemistry, Manufacturing and Controls) which she has put in practice at several multinational Pharmaceutical companies and at her own consultancy firm PHI for over 25 years now.
Regulatory Affairs
98
Technologie & Quality Affairs
85
Pharmaceutical Development
85